|
3/30/2023 0 Comments Cathode ray experiment explained
What do you mean by cathode? (1 Mrak)Īns. Magnetic and electric fields block them, and they have a negative charge. They travel straight and cast sharp shadows. They are formed in an evacuated tube through the cathode, or negative electrode, and move toward the anode. What are the properties of cathode rays? (1 Mrak)Īns. Thomson understood that the accepted model of an atom did not account for the particles charged positively or negatively. He proved that the cathode rays were charged negatively. Thomson invented the electron by playing with a tube that was Crookes, or cathode ray. How did JJ Thomson find the electron? (1 Mrak)Īns. If an evacuated tube of glass is fitted with two electrodes and a voltage is added, it is found that the glass opposite to the negative electrode glows from the electrons emitted by the cathode. Cathode rays are electron streams observed in vacuum tubes (also known as an electron beam or an e-beam). Where can you find a cathode ray tube? (1 Mrak)Īns. 
Cesium is used as a cathode because it releases electrons readily when heated or hit by light. What are cathode ray tubes made of? (1 Mrak)Īns. This also explains the electrically neutral nature of an atom. The number of electrons dispersed outside the nucleus is equal to the number of positively charged protons present in the nucleus. In a nutshell we came to know that the electrons are the basic constituent of all the atoms. And the significant observation that he made was that the characteristics of cathode rays or electrons did not depend on the material of electrodes or the nature of the gas present in the cathode ray tube. This theory helped physicists further to understand the atomic structure. Thomson concluded that rays are negatively charged particles present or moving around in a set of a positive charge. Summary: After completing the experiment J.J. These rays consisted of electrons, material particles and it caused various other materials to glow on striking them. This behaviour was similar to that of a negatively charged particle and this suggested that the rays contained electrons. The cathode rays were deflected by electrical and magnetic fields. Cathode rays originated from the cathode and travelled in straight lines carrying a negative charge. Thomson studied the characteristics and the constituents of cathode rays.Ĭertain properties of these cathode rays were concluded so let us discuss them now. These rays were named as the cathode rays.Įxperiments on the discharge tube were performed first in 1878 by an English physicist, William Crookes. 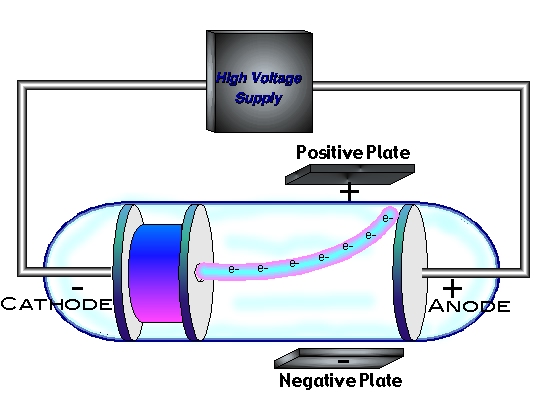
Due to this current, a stream of rays spreading from the cathode passed through the tube. In the discharge tube experiment at a high voltage with low pressure, an electrical current was passed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
